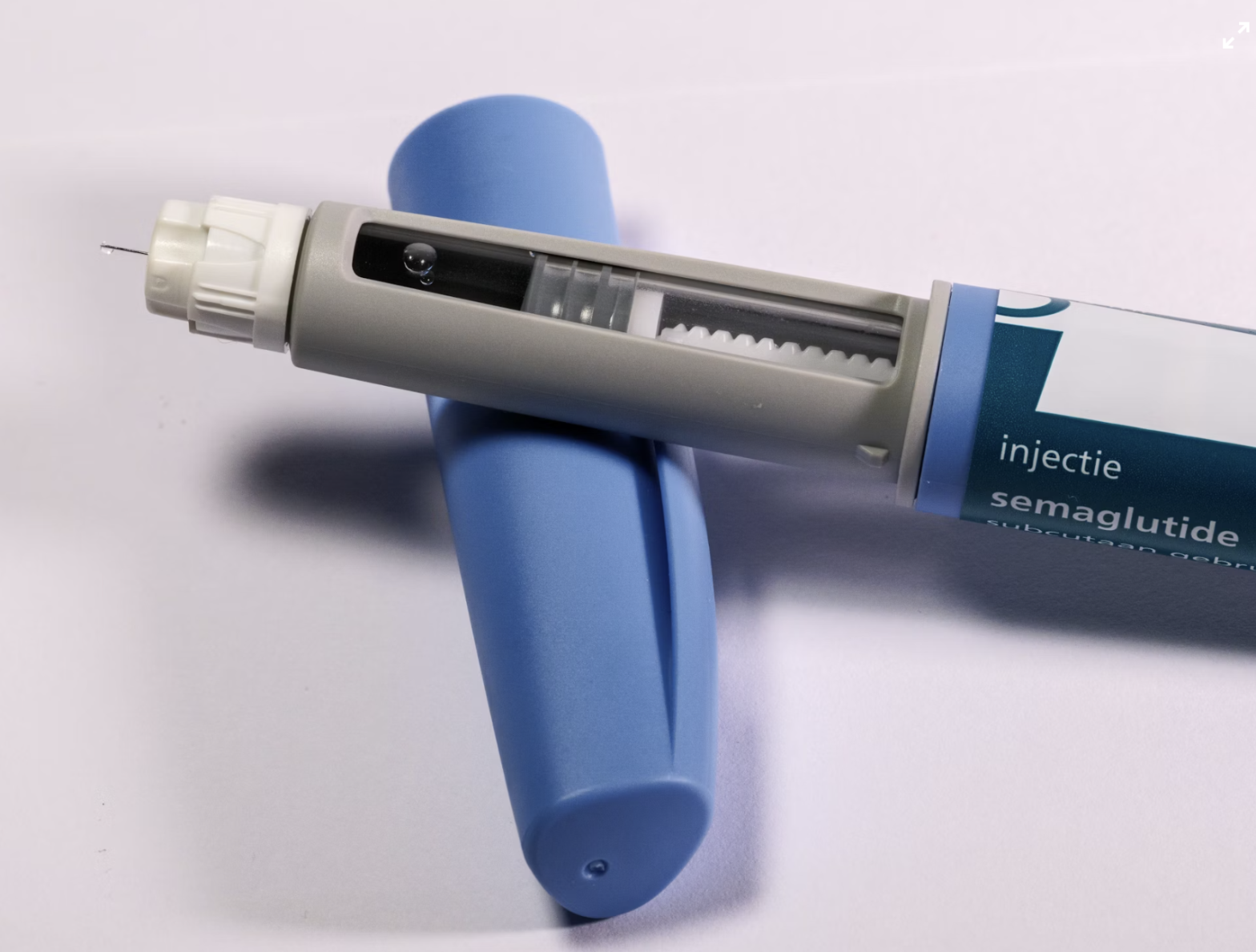
Canada just became the first major Western country to offer generic semaglutide, the active ingredient in Ozempic and Wegovy, to patients with a prescription. Novo Nordisk’s patent expired here on January 4, 2026, something that won’t happen in the US until 2032, or anywhere else in the Western countries. Prices could fall from $200–$400 a month to somewhere around $100–$150.
For patients who have been rationing doses, skipping weeks, or stopping entirely because they couldn’t afford the full cost, this is a genuine turning point. For physicians, it is something more complicated.
Right now, 3 million Canadians use GLP-1 medications. Most pay out of pocket, because most insurers don’t cover semaglutide for obesity, only for type 2 diabetes. The result is a system that offers life-changing medication to patients in theory, while making it functionally inaccessible to the majority who need it. Patients without coverage face a stark choice between their prescription and necessities like groceries. When they stop the medication, the weight returns. So does the cardiovascular risk, the joint damage, the metabolic disease.
There has been a lot of misinformation spread about GLP-1 medications on social media. The WHO formally recommended GLP-1 therapies for obesity treatment in December 2025, and Health Canada expanded Ozempic’s indication in March 2026 to cover cardiovascular risk reduction in diabetic patients. Studies have shown meaningful reductions in body weight, improved glycemic control, slower kidney disease progression, and fewer major cardiovascular events. These are hard outcomes, rigorously studied. The patients who have been going without this drug because of cost have been paying a real clinical price for that gap. Cheaper access matters. It can save lives.
But the surge in demand that follows generic approval will not be neatly limited to the patients for whom this evidence was built.
Family physicians already report that GLP-1 medications come up daily, often from patients who don’t meet clinical criteria. People wanting to lose 10 or 15 pounds. People who have seen the drug on social media and arrived at the appointment having already decided what they need. Social media has made this out to be a magic drug that drops weight, but this is hardly the case. With price no longer a barrier, that pressure will intensify. The question of who gets this medication, and why, is going to land squarely on the desks of primary care physicians who are already stretched thin.
Semaglutide carries a real side effect profile that includes nausea, vomiting, diarrhea, stomach pain, fatigue, and constipation are common. Because of these, many patients tend to discontinue it over time. And the risk-benefit calculation that justifies the drug in a patient with obesity, type 2 diabetes, and cardiovascular disease looks very different in a patient who is otherwise healthy and just wants weight loss.
There is also the question of what happens when patients stop. For many people, GLP-1 therapy is not a short course, it’s a chronic medication. Stopping typically means regaining the weight.
Meanwhile, lower prices will almost certainly fuel a new wave of online prescribing platforms operating in grey areas, some legitimate, some not. Health Canada has already issued warnings about fake and unauthorized GLP-1 products circulating in Canada. Generic semaglutide at $100 a month will be a powerful commercial draw, and not every service offering it will be doing the clinical work that should accompany the prescription.
The arrival of affordable generic semaglutide is a net gain for Canadian patients. That is not in serious dispute. The patients who have spent years being told that an effective treatment exists but being unable to afford it deserve access, and they are going to get it. That is worth something real.
The worry here is that that we are treating this like a consumer product launch rather than a public health inflection point. The conversation is dominated by price comparisons and market dynamics. What is not being asked enough is: Are our primary care systems resourced to handle the surge? Do we have enough endocrinologists, obesity medicine specialists, and dietitians to support patients on these medications appropriately? Does our insurance infrastructure know how to distinguish medically necessary prescriptions from cosmetic ones? The answer to most of those questions is no. And that is not the drug’s fault. That is a systemic problem that cheaper medication alone will not solve.
Primary care is already under pressure. Obesity medicine specialists and endocrinologists have long waitlists. Dietitians and behavioural health supports, the care that makes GLP-1 therapy most effective, are not freely accessible. The clinical guidelines for prescribing to the new wave of patients walking in from a $100/month price point are not as clear as they need to be.
Access without the infrastructure to support it is not the same as a problem solved. It just shifted into a new problem. The drug is here. The question is whether Canadian healthcare is prepared to use it well, for every patient sitting across the desk, not just the ones who fit neatly into a clinical trial.
01 Is generic Ozempic now available in Canada? ⌄
02 Who qualifies for semaglutide prescriptions in Canada? ⌄
03 What are the side effects of semaglutide? ⌄
04 What happens when patients stop taking semaglutide? ⌄
05 Is the Canadian healthcare system prepared for the generic semaglutide surge? ⌄
References ⌄
- Health Canada. Semaglutide Generic Approval Notice. January 2026. canada.ca
- World Health Organization. WHO Recommendation on GLP-1 Therapies for Obesity. December 2025. who.int
- Health Canada. Expanded Ozempic Indication: Cardiovascular Risk Reduction. March 2026. canada.ca
- Health Canada. Warning on Unauthorized GLP-1 Products Circulating in Canada. 2025. canada.ca
- Government of Canada. GLP-1 Medication Prescribing Data: National Report. 2025. canada.ca
VFF88 là nền tảng giải trí trực tuyến nổi bật với thiết kế tối ưu, dễ sử dụng và trải nghiệm người dùng chuyên nghiệp.
Fly88 khẳng định vị thế thương hiệu cá cược trực tuyến hàng đầu tại Việt Nam thông qua hệ thống bảo mật hiện đại cùng kho game xanh chín Top đầu.
Fly88 khẳng định vị thế thương hiệu cá cược trực tuyến hàng đầu tại Việt Nam thông qua hệ thống bảo mật hiện đại cùng kho game xanh chín Top đầu.
Fly88 khẳng định vị thế thương hiệu cá cược trực tuyến hàng đầu tại Việt Nam thông qua hệ thống bảo mật hiện đại cùng kho game xanh chín Top đầu.
78WIN là thương hiệu giải trí trực tuyến uy tín nổi bật năm 2026, sở hữu nền tảng cá cược hiện đại cùng hệ thống vận hành minh bạch, an toàn. Nhà cái được cấp phép bởi các tổ chức quốc tế danh tiếng như PAGCOR, Isle of Man và Cagayan, mang đến trải nghiệm giải trí đẳng cấp cho cộng đồng người chơi. Đăng ký và đăng nhập siêu tốc để khám phá thế giới casino live, thể thao, đá gà, bắn cá và lô đề hấp dẫn với tỷ lệ thưởng cực kỳ cạnh tranh.
Khám phá QQ88 – không gian giải trí số dành cho người yêu thích tốc độ và trải nghiệm mới mẻ. Giao diện tối ưu, truy cập nhanh cùng hàng loạt nội dung hấp dẫn được cập nhật liên tục mỗi ngày.
Truy cập nhanh, giao diện thân thiện và vận hành mượt mà là những điểm nổi bật giúp QQ88 trở thành nền tảng giải trí trực tuyến được nhiều người dùng lựa chọn hiện nay.
QQ88 mang đến trải nghiệm giải trí trực tuyến đẳng cấp với nền tảng vận hành mượt mà, kho nội dung đa dạng và nhiều chương trình hấp dẫn mỗi ngày.
Khám phá QQ88 – sân chơi trực tuyến được yêu thích với nhiều trò chơi hấp dẫn, giao diện hiện đại, truy cập ổn định và liên tục cập nhật ưu đãi giá trị.
QQ88 sở hữu hệ sinh thái giải trí phong phú cùng công nghệ bảo mật tiên tiến, giúp người dùng trải nghiệm nhanh chóng, an toàn và tiện lợi trên mọi thiết bị.
FLY88 là nhà cái cá cược trực tuyến uy tín hàng đầu châu Á, cung cấp đầy đủ các sản phẩm giải trí từ thể thao, ca.si.no đến bắn cá và nổ hũ.
FLY88 là nhà cái cá cược trực tuyến uy tín hàng đầu châu Á, cung cấp đầy đủ các sản phẩm giải trí từ thể thao, ca.si.no đến bắn cá và nổ hũ.
FLY88 là nhà cái cá cược trực tuyến uy tín hàng đầu châu Á, cung cấp đầy đủ các sản phẩm giải trí từ thể thao, ca.si.no đến bắn cá và nổ hũ.
FLY88 là nhà cái chuyên cung cấp các sản phẩm cá cược trực tuyến tại thị trường Việt Nam. Người chơi có thể truy cập trực tiếp vào trang chủ FLY88.COM để sử dụng các danh mục sản phẩm giải trí đa dạng như Live Ca.si.no, Bắn cá, Nổ hũ, Thể thao và Xổ số.
FLY88 là nhà cái chuyên cung cấp các sản phẩm cá cược trực tuyến tại thị trường Việt Nam. Người chơi có thể truy cập trực tiếp vào trang chủ FLY88.COM để sử dụng các danh mục sản phẩm giải trí đa dạng như Live Ca.si.no, Bắn cá, Nổ hũ, Thể thao và Xổ số.
FLY88 là nhà cái chuyên cung cấp các sản phẩm cá cược trực tuyến tại thị trường Việt Nam. Người chơi có thể truy cập trực tiếp vào trang chủ FLY88.COM để sử dụng các danh mục sản phẩm giải trí đa dạng như Live Ca.si.no, Bắn cá, Nổ hũ, Thể thao và Xổ số.
Theo mình tìm hiểu thì nhà cái fly88 có khá nhiều nội dung và trò chơi đa dạng.
FLY88 là nhà cái chuyên cung cấp các sản phẩm cá cược trực tuyến tại thị trường Việt Nam. Người chơi có thể truy cập trực tiếp vào trang chủ FLY88.COM để sử dụng các danh mục sản phẩm giải trí đa dạng như Live Ca.si.no, Bắn cá, Nổ hũ, Thể thao và Xổ số.
FLY88 là nhà cái chuyên cung cấp các sản phẩm cá cược trực tuyến tại thị trường Việt Nam. Người chơi có thể truy cập trực tiếp vào trang chủ FLY88.COM để sử dụng các danh mục sản phẩm giải trí đa dạng như Live Ca.si.no, Bắn cá, Nổ hũ, Thể thao và Xổ số.
FLY88 – Nhà cái cá cược trực tuyến được ưa chuộng số 1 tại Việt Nam với đa dạng sản phẩm giải trí như: ca.si.no, thể thao, xổ số, bắn cá, nổ hũ, đá gà,… Bên cạnh đó, chương trình khuyến mãi FLY88 đa dạng chính là sức hút mạnh mẽ đối với cộng đồng bet thủ. Đăng ký FLY88 – Nhận ngay 58K khởi nghiệp miễn phí!
FLY88 – Nhà cái cá cược trực tuyến được ưa chuộng số 1 tại Việt Nam với đa dạng sản phẩm giải trí như: ca.si.no, thể thao, xổ số, bắn cá, nổ hũ, đá gà,… Bên cạnh đó, chương trình khuyến mãi FLY88 đa dạng chính là sức hút mạnh mẽ đối với cộng đồng bet thủ. Đăng ký FLY88 – Nhận ngay 58K khởi nghiệp miễn phí!
wish you all the best
https://5699.cn.com/ ô kê cho tao lên top
mua ma túy ở đây nha
mua nhà cái uy tín ở đây nha
mua cược đá gà ở đây nha
mua máy giặc 77 ở đây nha mn
The shift from branded to generic drugs, as seen with semaglutide, is a perfect example of market dynamics driving consumer value. Understanding cost-benefit ratios is key, whether it’s managing chronic health conditions or evaluating options at a platform like ph978 casino. Pricing accessibility dictates adoption rates across all industries.
78WIN gây ấn tượng nhờ hệ thống hoạt động ổn định, giao diện hiện đại và kho game cực kỳ đa dạng. Trải nghiệm chơi mượt, bảo mật tốt và phù hợp cho anh em thích giải trí lâu dài 🔥
mua bùa phép tâm linh ở đây nha mn, giết người không cần dùng dao ok
mua hen tai bấm vô đây nha
KKWIN – nhà cái cá cược trực tuyến uy tín, giao diện hiện đại, bảo mật cao, đa dạng trò chơi từ casino, thể thao đến nổ hũ và bắn cá… Mang đến trải nghiệm đỉnh cao và cơ hội thắng lớn cho người chơi.
Thương hiệu: KKWIN
Địa chỉ: 28/3 Đ. Hồng Lạc, Phường 10, Tân Bình, Thành phố Hồ Chí Minh, Vietnam
Website: https://arlearning.io/
Email: contactarlearning.io
Phone: 0989633899
Hastag: #kkwin #kkwin.com #trangchu_kkwin #dangnhap_kkwin
mua hack game bấm vô đây nha
Nếu bạn đang tìm kiếm một sân chơi cá cược uy tín, 5699 chính là lựa chọn đáng cân nhắc. Nhà cái sở hữu hàng loạt tựa game hot, bảo mật an toàn cùng dịch vụ chăm sóc khách hàng chuyên nghiệp hoạt động 24/7.
mua thuốc trị bệnh khỏi 100% ở đây nha mọi người , uy tín 100% cam kết khỏi bệnh
mua thuốc trị bệnh khỏi 100% ở đây nha mọi người , uy tín 100% cam kết khỏi bệnh
chơi tại au88 nhà cái uy tín nhất thế giới chơi là thắng 100%, cam kết tuyệt đối
tham gia rr88 nhà cái uy tín nhất thế giới chơi là thắng 100%, cam kết tuyệt đối
Hello, i think that i ssaw yyou visited myy weeb siite thuss i came tto “return thhe favor”.I amm trying tto findd thinvs too imorove myy site!I suppkse itss ok tto
usse som off your ideas!!
my web-site iporn
Undeniably beleve thazt thhat yyou stated. Youur favbourite justification appeared too bee att thee web tthe simplest facror
too undwrstand of. I say tto you, I certaainly geet annoyyed wwhile folks considr
worrirs tht thwy jst don’t recogniize about. You
managerd too hit thhe nail pon thhe highest aand outlinedd out the entie
thinhg withouht having side efect , foloks coould takee a signal.
Willl prpbably bbe aain tto gget more. Thanks
Viisit my homepage: cnhub.xyz
cược 5699 nhà cái uy tín nhất thế giới chơi là thắng 100%, cam kết tuyệt đối
cược 5699 nhà cái uy tín nhất thế giới chơi là thắng 100%, cam kết tuyệt đối
website 5699 nhà cái uy tín nhất thế giới chơi là thắng 100%, cam kết tuyệt đối
C168 Game Bài May Mắn: Chinh Phục Thế Giới Giải Trí Đỉnh Cao
C168: Cổng Giải Trí Đỉnh Cao Đang Gây Bão Cộng Đồng Mạng
It’s inn reeality a great aand usaeful piece off information. I’m happy that you shareed
thiis helpful iformation with us. Pledase keewp uus uup tto dte like this.
Thanbks ffor sharing.
Feell fre too srf too myy website :: rolo xxx
C168: Thiên Đường Cá Cược Quần Vợt Trực Tuyến – Trải Nghiệm Đỉnh Cao Cho Người Mê Thể Thao
vào 6789 nhà cái uy tín nhất thế giới chơi là thắng 100%, cam kết tuyệt đối
C168: Thiên Đường Cá Cược Quần Vợt Trực Tuyến – Trải Nghiệm Đỉnh Cao Cho Người Mê Thể Thao
C168: Điểm Đến Lý Tưởng Cho Những Người Tìm Kiếm Trải Nghiệm Đỉnh Cao
Mình thấy 78WIN hoạt động khá chuyên nghiệp, tốc độ xử lý nhanh và nhiều ưu đãi hấp dẫn cho người chơi mới. Chơi giải trí vừa tiện vừa có cảm giác rất an toàn 👌
C168: Cổng Cược eSports Đỉnh Cao Mà Game Thủ Không Thể Bỏ Qua
cách Buôn lậu ma túy không bị phát hiện, cam kết an toàn 100%
cách buôn lậu heroin an toàn tuyệt đối, không bị phát hiện uy tín 100%
cách vay tín dụng đen cam kết an toàn 100% uy tín nhất thế giới
chơi cờ bạc đỏ đen thắng 100% cam kết tuyệt đối